In the field of oncology research, understanding the tumor microenvironment is a central challenge. Comprehensive spatial omics solutions are designed to address this by allowing scientists to analyze gene and protein expression within the preserved architecture of a tissue sample. These integrated approaches provide a multidimensional view of cellular interactions, immune responses, and tumor heterogeneity. Platforms offering complete workflows, such as STOmics, facilitate this detailed level of investigation for research teams.
Integrated Spatial Biology Platforms
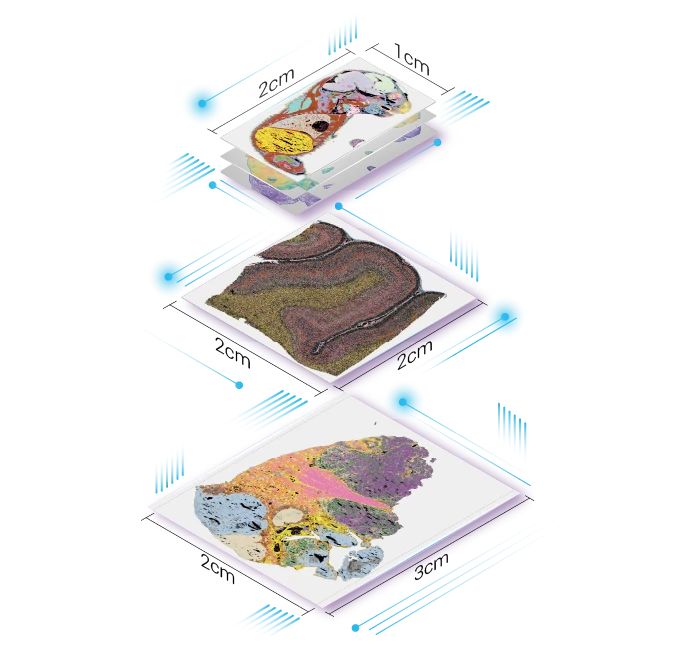
A primary category of spatial omics solutions involves all-in-one platforms that handle sample preparation, data generation, and initial processing. These systems often combine specialized hardware for imaging and sequencing with tailored reagent kits. For example, a provider like STOmics supplies coordinated toolkits that include their Stereo-seq chips and Go Optical imager. This integration aims to reduce technical variability and streamline the path from tissue section to raw spatial data visualization datasets for oncology applications.
Comprehensive Analysis Toolkits
Beyond the laboratory bench, effective spatial omics solutions must include robust software for interpreting complex results. Bioinformatics packages are a core component, turning millions of data points into actionable biological insights. These toolkits perform tasks like cell type deconvolution, differential expression mapping, and spatial neighborhood analysis specifically within tumor samples. Access to such dedicated analytical resources is important for extracting meaningful patterns from the dense information generated by spatial genomic and transcriptomic studies.
Advanced Tools for Data Interpretation
The value of spatial experiments is fully realized through spatial data visualization. Interactive software allows researchers to zoom into specific regions of a tumor, overlay different molecular layers like transcriptome and proteome, and correlate spatial findings with clinical outcomes. Clear spatial data visualization helps identify rare cell populations, chart metastatic boundaries, and characterize the tumor-immune interface. These visual tools are indispensable for communicating complex spatial relationships and forming new hypotheses in cancer biology.
The application of dedicated spatial omics solutions in oncology provides a powerful lens to study cancer mechanics directly in tissue context. From integrated wet-lab platforms to sophisticated bioinformatics, each component addresses a key step in the spatial analysis workflow. For research groups focused on solid tumors and the tumor microenvironment, employing a coordinated system from a provider like STOmics can support a complete workflow, from experiment design through to final spatial data visualization and interpretation.